FBP2 (1-339, His-tag) Human Protein
CAT#: AR51990PU-N
FBP2 (1-339, His-tag) human protein, 0.1 mg
Size: 20 ug
|
Need it in bulk or customized? Get a free quote |
CNY 9,910.00
货期*
详询
规格
Specifications
| Product Data | |
| Species | Human |
| Expression Host | E. coli |
| Expression cDNA Clone or AA Sequence |
MGSSHHHHHH SSGLVPRGSH MGSMTDRSPF ETDMLTLTRY VMEKGRQAKG TGELTQLLNS MLTAIKAISS AVRKAGLAHL YGIAGSVNVT GDEVKKLDVL SNSLVINMVQ SSYSTCVLVS EENKDAIITA KEKRGKYVVC FDPLDGSSNI DCLASIGTIF AIYRKTSEDE PSEKDALQCG RNIVAAGYAL YGSATLVALS TGQGVDLFML DPALGEFVLV EKDVKIKKKG KIYSLNEGYA KYFDAATTEY VQKKKFPEDG SAPYGARYVG SMVADVHRTL VYGGIFLYPA NQKSPKGKLR LLYECNPVAY IIEQAGGLAT TGTQPVLDVK PEAIHQRVPL ILGSPEDVQE YLTCVQKNQA GS
|
| Tag | His-tag |
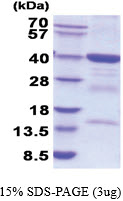
| Predicted MW | 39.0 kDa |
| Concentration | lot specific |
| Purity | >90% by SDS - PAGE |
| Buffer | Presentation State: Purified State: Liquid purified protein Buffer System: 20 mM Tris-HCl buffer (pH 8.0) containing 0.15M NaCl, 10% glycerol, 1 mM DTT |
| Preparation | Liquid purified protein |
| Storage | Store undiluted at 2-8°C for one week or (in aliquots) at -20°C to -80°C for longer. Avoid repeated freezing and thawing. |
| Stability | Shelf life: one year from despatch. |
| Bioactivity | Specific: Specific activity is > 1,500 pmol/min/ug obtained by measuring the increase of NADPH in absorbance at 340 nm resulting from the reduction of NADP. One unit will oxidize 1.0 pmole of fructose 1,6 diphosphate to fructose 6-phosphate and inorganic phosphate per minute at pH 9.5 at 37C. |
| Reference Data | |
| RefSeq | NP_003828 |
| Locus ID | 8789 |
| UniProt ID | O00757 |
| Cytogenetics | 9q22.32 |
| Summary | This gene encodes a gluconeogenesis regulatory enzyme which catalyzes the hydrolysis of fructose 1,6-bisphosphate to fructose 6-phosphate and inorganic phosphate. [provided by RefSeq, Jul 2008] |
| Protein Families | Druggable Genome |
| Protein Pathways | Fructose and mannose metabolism, Glycolysis / Gluconeogenesis, Insulin signaling pathway, Metabolic pathways, Pentose phosphate pathway |
Documents
| FAQs |
| SDS |
Customer
Reviews
Loading...


 United States
United States
 Germany
Germany
 Japan
Japan
 United Kingdom
United Kingdom
 China
China